Trends...
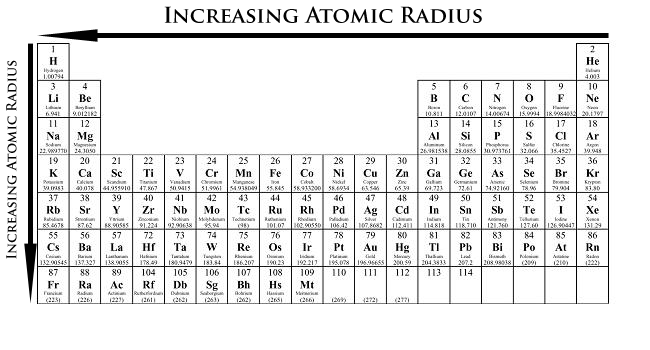
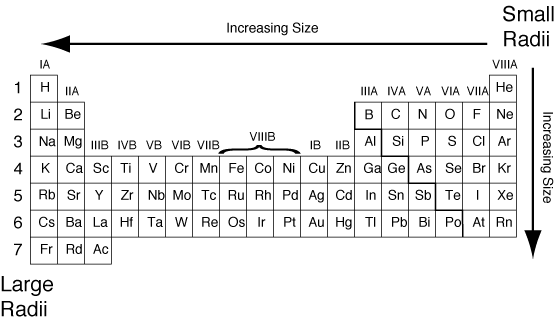
Atomic radius= size of atom from the nucleus to outer shell
- atomic radius increases from right to left
- size of atom increases from top to bottom
Ionic radius= radius of an atom's ion
- As you move down the periodic table the ionic radius increases
- As you move across the periodic table (to the right) the ionic radius decreases
- As you move down the periodic table the ionic radius increases
- As you move across the periodic table (to the right) the ionic radius decreases
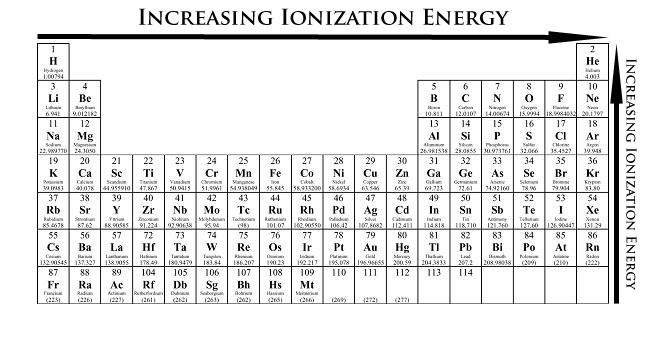
Ionization energy= energy required to remove an electron to form a positive ion
- As you move up the ionization energy increases
- As you move to the right the ionization energy increases as well
Tip: Think of somebody walking up a hill, you need to use more energy to walk up and keep walking forward
- As you move up the ionization energy increases
- As you move to the right the ionization energy increases as well
Tip: Think of somebody walking up a hill, you need to use more energy to walk up and keep walking forward
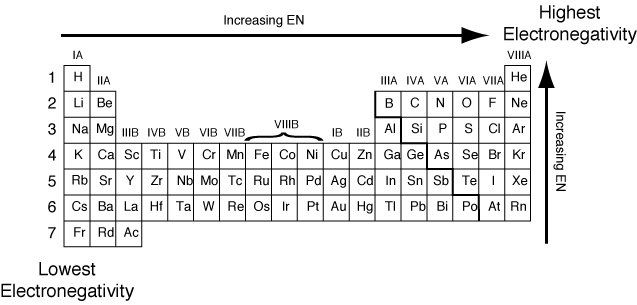
Electronegativity=
a measure of the tendency of an atom to attract a bonding pair of
electrons
- Electronegativity increases as you move to the far right corner of the periodic table
- Electronegativity increases as you move to the far right corner of the periodic table